|
Some elements are called metalloids because they are in between metals and non-metals, and you can see that the metalloids are also in between the metals and non-metals in the periodic table. Non-metals don't conduct, and are often softer or easier to break than metals. 
Metals are shiny, and they conduct heat and electricity. Notice how metals are on the left and bottom of the periodic table, while non-metals are on the right and top. To learn your way around the table, try going to this much fancier periodic table. (lanthanoides, actinoides) similar to each other, most have M 3+ ionic form Metals with multiple valences and ionic forms, initially hard to fit into periodic table, many exist as M 2+ Very unreactive monatomic gases, valence 0 Non-metals, very reactive, volatile elements, valence 1, usually X – ions Soft, less reactive metals, valence 2, almost always M 2+ ions Soft, extremely reactive metals, valence 1, almost always M + ions Here is some info about the important groups. To make these predictions, you will need to know a little about the different families or groups, which are the columns of the table. How do you use the periodic table? It can help you predict many important properties of elements. You can search for an element by name or symbol and it will be highlighted so it's easy to find. If you point at a symbol, it will show the element name. 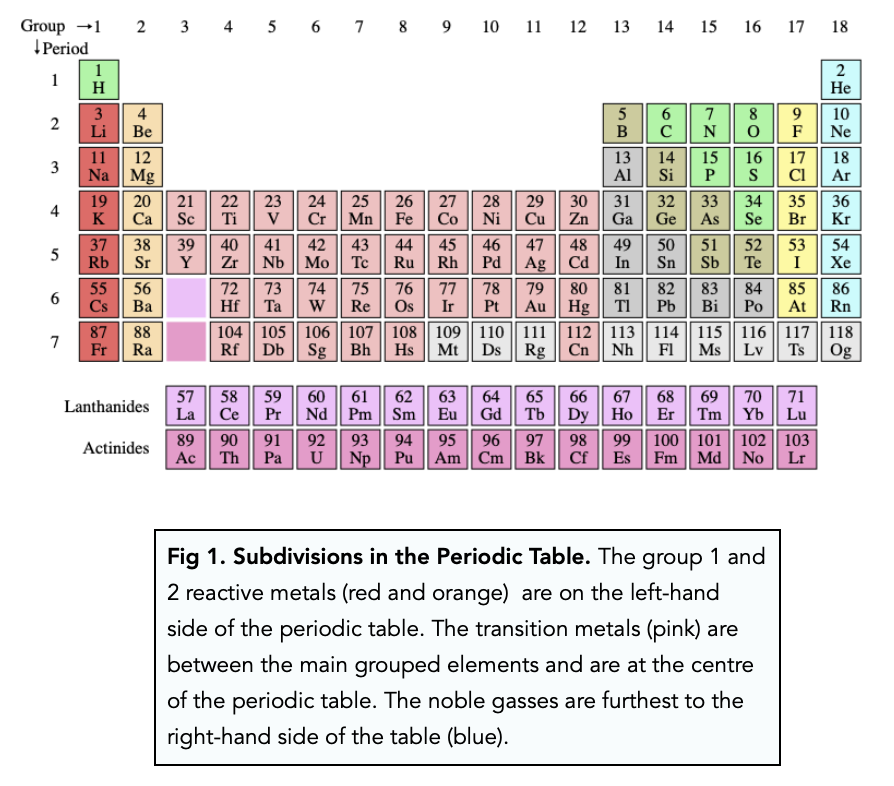
It shows the symbols, atomic numbers, and average atomic masses for each element. 
The Periodic Table is an essential tool for chemists. Associate family name to description (especially for groups 1 - 2 and 16 - 18).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
